Bacterial sugars in the gut could be a hidden contributor to two of the most serious neurodegenerative diseases that afflict the brain, new research suggests.
Amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) are closely linked conditions that sometimes overlap, and both involve the death of neurons.
This affects muscle movement in ALS (which is eventually fatal), and leads to major changes in behavior and language in FTD, which can also sometimes affect movement.
It's still not clear how either disease gets started, and here a team led by researchers from Case Western Reserve University in the US wanted to see if gut bacteria might play a role, based on previous findings that suggested a link with ALS.
Using a mouse model, the research turned up a specific type of glycogen produced by gut bacteria. It seems this sugar triggers brain inflammation and neuron death.
"We found that harmful gut bacteria produce inflammatory forms of glycogen – a type of sugar – and that these bacterial sugars trigger immune responses that damage the brain," says Aaron Burberry, an assistant professor of pathology at Case Western Reserve University.
Now we know that, we can start working on developing treatments to target it.
Both ALS and FTD can be caused by a particular variation of the C9ORF72 gene, but not everyone who has this variation develops the diseases.
The main impetus behind the research was to figure out what other triggers may be contributing among those with the gene variant.
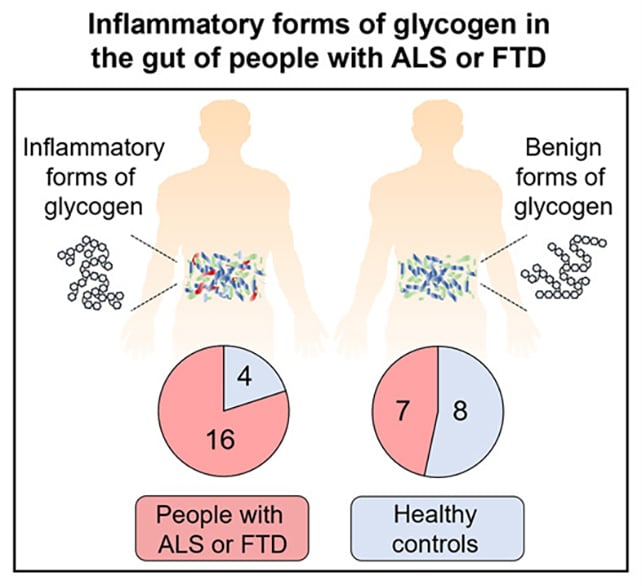 The researchers linked inflammatory glycogen to ALS and FTD. (McCourt et al., Cell Rep., 2026)
The researchers linked inflammatory glycogen to ALS and FTD. (McCourt et al., Cell Rep., 2026)The researchers were methodical in their approach. They engineered mice without the C9ORF72 gene to mimic the variant in people, then tested a variety of gut bacteria mixes in the mice to see how their immune systems would react.
That led the team to glycogen production, and from there to Parabacteroides merdae bacteria, one of the strains that creates it. When this bacteria was introduced to mice bred with no gut bacteria, it caused serious inflammation and a breakdown of the blood-brain barrier.
Further tests on human stool samples showed higher than normal levels of inflammatory glycogen in 15 out of 22 ALS patients and a single FTD patient, but in only four out of 12 healthy controls.
The thinking is that when the body detects the potentially dangerous sugar, the immune system kicks into overdrive, which then starts to affect the brain. Importantly, the protein encoded by C9ORF72 seems to act as a brake on glycogen – which is where the problem with the gene variation comes in.
"Our demonstration that microbes that accumulate inflammatory forms of glycogen are enriched in the gut of ALS patients suggests that microbial glycogen may be an important example among many environmental and lifestyle factors that interact with predisposing genotypes to contribute risk of ALS onset and progression," write the researchers in their published paper.
One of the most positive results from the research is that when affected mice were given an enzyme called alpha-amylase – which breaks down glycogen – inflammation levels were reduced and their lifespans were extended. However, it did not improve their motor performance.
That suggests that one day we may be able to develop treatments that could interrupt this inflammatory chain reaction, targeting the gut rather than the brain, though it's early days for that aspect of the research.
Related: One in Three Dementia Cases Is Linked to Disease Outside The Brain
Scientists are discovering more and more about the ways our guts and our brains are connected, especially when it comes to health, and this new study adds to that.
Next, the researchers want to move the investigation beyond mouse models, looking in more detail at human participants and different types of glycogen-producing bacteria.
"To understand when and why harmful microbial glycogen is produced, the team will next conduct larger studies surveying gut microbiome communities in ALS/FTD patients before and after disease onset," says Burberry.
"Clinical trials to determine whether glycogen degradation in ALS/FTD patients could slow disease progression are also supported by our findings and could begin in a year."
The research has been published in Cell Reports.

.jpg) 3 hours ago
3
3 hours ago
3

 English (US)
English (US)